
T
here are now numerous non sterile disinfectant wipes available, predominantly for infection control use in healthcare environments. With the changes to the transfer disinfection requirements for Specials Manufacturers in the UK[1] and unlicensed aseptic units[2]; now including a sporicidal wiping phase there are also more sporicidal disinfectant wipes available.However, as previously discussed[3] ; how does a customer ensure that a presaturated wipe works as well as the ready to use fluids as there are known problems with adding certain disinfectant active ingredients to certain wipe or mop substrates[4,5,6,7].
Incompatibility between the disinfectant and the substrate.
When the previous test work was carried out there was no specific European test for presaturated wipes. We utilised the existing standard EN suspension and surface tests on eluate extracted from the wipe at the end of shelf life to show that the wipe being in contact with the fluid had not had any adverse effect on the efficacious of the disinfectant. When evaluating presaturated wipes it is crucial to ensure that efficacy testing has been carried out on fluid extracted from the wipe, otherwise as the studies have shown the fabric can have had an unknown detrimental effect on the disinfectant which worked perfectly well as a ready-to-use fluid.
Work was carried out on a range of sterile and non sterile presaturated wipes currently on the market.[8] Wipes at various points within their shelf life were squeezed to a standardised method to remove as much fluid as possible from the wipe and then the eluate was tested against the standard EN disinfectant tests, EN 1276 for bacterial efficacy[9], EN1650 for fungal efficacy[10] and EN 13704 for sporicidal efficacy[11]. Table 1 shows the results obtained and where the wipe eluate didn’t reach the required level of efficacy as claimed for the equivalent fluid product. The contact time to prove sporicidal efficacy against EN 13704 is 60 mins. However, as this doesn’t tend to be practical in-use, many companies test at a shorter contact time of between 3 – 15 mins. The test work was carried out at 15 mins which is a longer contact time than claimed for any of the products tested.
Work was carried out on a range of sterile and non sterile presaturated wipes currently on the market.[8] Wipes at various points within their shelf life were squeezed to a standardised method to remove as much fluid as possible from the wipe and then the eluate was tested against the standard EN disinfectant tests, EN 1276 for bacterial efficacy[9], EN1650 for fungal efficacy[10] and EN 13704 for sporicidal efficacy[11]. Table 1 shows the results obtained and where the wipe eluate didn’t reach the required level of efficacy as claimed for the equivalent fluid product. The contact time to prove sporicidal efficacy against EN 13704 is 60 mins. However, as this doesn’t tend to be practical in-use, many companies test at a shorter contact time of between 3 – 15 mins. The test work was carried out at 15 mins which is a longer contact time than claimed for any of the products tested.
Table 1:
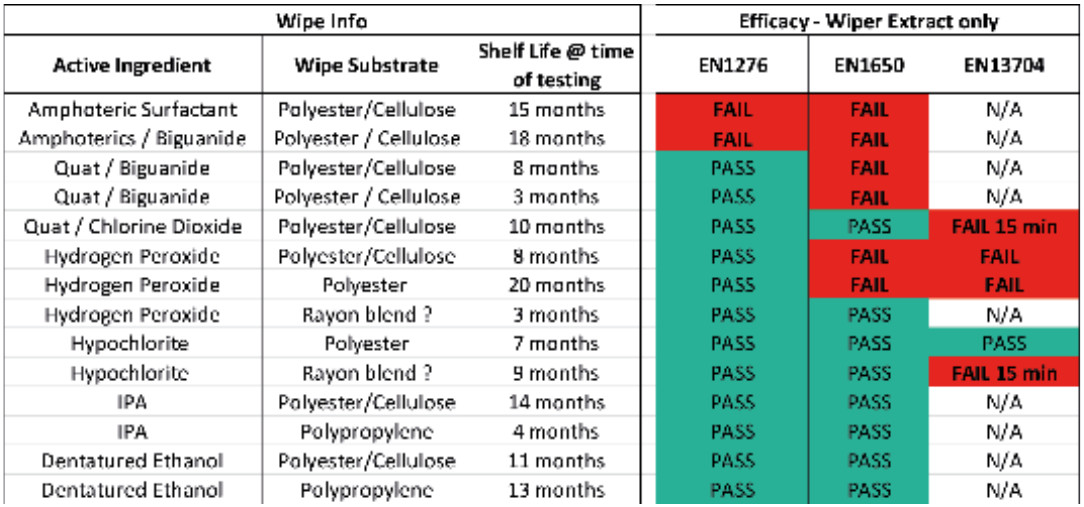
As can be seen from these results many of the wipes tested didn’t meet the claims made for the equivalent RTU fluid. The tests were performed twice to eliminate any possible error in the testing (all tests did pass validation).
It was concluded that the wipes had probably failed for a number of reasons. Hydrogen peroxide is an equilibrium product where contact with certain materials is likely to start the decomposition process, known as “gassing off”. Hydrogen peroxide breaks down to water and oxygen, this decomposition process can leave a wipe with little to no activity and a pouch overfilled with air (oxygen). Additional testing showed that the amphoteric and quaternary ammonium based wipes had a significant portion of the active ingredient binding into the wipe and not being laid down onto the surface.
It was concluded that the wipes had probably failed for a number of reasons. Hydrogen peroxide is an equilibrium product where contact with certain materials is likely to start the decomposition process, known as “gassing off”. Hydrogen peroxide breaks down to water and oxygen, this decomposition process can leave a wipe with little to no activity and a pouch overfilled with air (oxygen). Additional testing showed that the amphoteric and quaternary ammonium based wipes had a significant portion of the active ingredient binding into the wipe and not being laid down onto the surface.
A specific test for wipes ?
It seemed apparent that with the increase in availability and use of presaturated wipes that there was a need for a specific EN test which was specifically designed to test wipes. The current tests available for testing disinfectants unless carried out on the eluate as above are not able to be modified to allow the testing of wipes. The closest surface test, BS EN 13697:2015 Chemical disinfectants and antiseptics. Quantitative non-porous surface test for the evaluation of bactericidal and/ or fungicidal activity of chemical disinfectants used in food, industrial, domestic and institutional areas. (phase 2, step 2) does not include any mechanical action.
There are two international test norms designed to test presaturated wipes. ASTM E2362 — 15 “Standard Practice for Evaluation of Presaturated or Impregnated Towelettes for Hard Surface Disinfection” which is used to substantiate disinfectant wipe claims by the EPA. Contaminated test surfaces are wiped with the presaturated wipe. Either 10 or 60 contaminated test surfaces are treated with the wipe. After incubation, the number of tubes showing growth of the target microorganism is recorded. To “pass” a 60 carrier test, at least 59 of the 60 surfaces wiped with the towelettes must demonstrate complete disinfection (no detectable growth of the target microorganism in the test tubes containing neutralizing growth medium). To “pass” a 10 carrier test, complete disinfection must take place on all of the wiped test surfaces.
This test still has some weaknesses; similarly, to the AOAC spray test there is variability on the basis of statistics alone, certain test parameters are not standardised (eg humidity levels during drying, concentration of bacteria on the test surface). Whilst closer to practice-mimicking conditions using one wipe to treat all surfaces with virtually all the fluid used up, would not be practical in a real world scenario and the test does not alleviate any variability in the pressure and technique used to wipe the test surface.
There are two international test norms designed to test presaturated wipes. ASTM E2362 — 15 “Standard Practice for Evaluation of Presaturated or Impregnated Towelettes for Hard Surface Disinfection” which is used to substantiate disinfectant wipe claims by the EPA. Contaminated test surfaces are wiped with the presaturated wipe. Either 10 or 60 contaminated test surfaces are treated with the wipe. After incubation, the number of tubes showing growth of the target microorganism is recorded. To “pass” a 60 carrier test, at least 59 of the 60 surfaces wiped with the towelettes must demonstrate complete disinfection (no detectable growth of the target microorganism in the test tubes containing neutralizing growth medium). To “pass” a 10 carrier test, complete disinfection must take place on all of the wiped test surfaces.
This test still has some weaknesses; similarly, to the AOAC spray test there is variability on the basis of statistics alone, certain test parameters are not standardised (eg humidity levels during drying, concentration of bacteria on the test surface). Whilst closer to practice-mimicking conditions using one wipe to treat all surfaces with virtually all the fluid used up, would not be practical in a real world scenario and the test does not alleviate any variability in the pressure and technique used to wipe the test surface.
A more recently introduced presaturated wipe test, ASTM E2967—15 — Standard Test Method for Assessing the Ability of Pre-wetted Towelettes to Remove and Transfer Bacterial Contamination on Hard, Non-Porous Environmental Surfaces using the Wiperator, attempts to remove some of this variability by using a reproducible mechanical action for wiping generated by the “Wiperator”. The Wiperator was developed by Filtaflex Ltd and the Centre for Research on Environmental Microbiology of Ottawa University (CREM).
It claims to greatly improves consistency compared with manual test methods by accurately controlling the applied force, rubbing speed and duration of wiping. During a test the wipe moves in an orbit of 10-mm dia at 1 rev/sec, also oscillating through 6 degrees of arc. Wiping continues for multiples of 5 sec, up to 45 sec. The default wipesample contact force is 150 g, which equates to a rubbing force of 800 g or more in a real-life wiping situation, an accessory 150 g weight permits testing 300 g forces. It is open to some debate whether in a cleanroom environment this does mimic the way and with what force a wipe is used.
Work carried out by Sattar et al [12] evaluated the new ASTM test to demonstrate efficacy of disinfectant and detergent wipes. Work was carried out by three independent laboratories on five types of commercially available presaturated wipe products. All five wipes (4 detergent wipes and 1 disinfectant wipe) achieved a greater than log 4 reduction against bacteria but only the disinfectant wipe prevented the transfer onto another surface. They concluded that this standard method ensured greater precision and reproducibility when testing presaturated wipes with mechanical action. However, this test has still not been in common usage in Europe for the testing of cleanroom presaturated wipes.
It claims to greatly improves consistency compared with manual test methods by accurately controlling the applied force, rubbing speed and duration of wiping. During a test the wipe moves in an orbit of 10-mm dia at 1 rev/sec, also oscillating through 6 degrees of arc. Wiping continues for multiples of 5 sec, up to 45 sec. The default wipesample contact force is 150 g, which equates to a rubbing force of 800 g or more in a real-life wiping situation, an accessory 150 g weight permits testing 300 g forces. It is open to some debate whether in a cleanroom environment this does mimic the way and with what force a wipe is used.
Work carried out by Sattar et al [12] evaluated the new ASTM test to demonstrate efficacy of disinfectant and detergent wipes. Work was carried out by three independent laboratories on five types of commercially available presaturated wipe products. All five wipes (4 detergent wipes and 1 disinfectant wipe) achieved a greater than log 4 reduction against bacteria but only the disinfectant wipe prevented the transfer onto another surface. They concluded that this standard method ensured greater precision and reproducibility when testing presaturated wipes with mechanical action. However, this test has still not been in common usage in Europe for the testing of cleanroom presaturated wipes.
EN16615:2015
Until recently there was no EN standard for assessing disinfectant wipe efficacy but EN16615[13] was launched in 2015 as a carrier test for establishing whether a wipe has a bactericidal or yeasticidal effect. It is designed for presaturated wipes but the wipe could be saturated with the chemical under test at point of use. There are options to include an interfering substance to replicate clean and dirty conditions. The contact time can also be varied from 1 min to 60 mins. Wiping is carried out with the wipe wrapped around a granite block weighing 2.3 – 2.5 kg to standardise the wiping procedure and to simulate the average pressure when wiping.
The block is wiped over 4 fields marked on a pvc sheet. The organism under test is dried onto field 1. The granite block is then passed over all the fields and back again in one single, fluid motion taking 2 seconds. In test field 1; a log 5 reduction needs to be achieved against bacteria or a log 4 reduction against yeasts. In fields 2 to 4 there needs to be an average carry-over of less than 50 cfu for each organism.
The block is wiped over 4 fields marked on a pvc sheet. The organism under test is dried onto field 1. The granite block is then passed over all the fields and back again in one single, fluid motion taking 2 seconds. In test field 1; a log 5 reduction needs to be achieved against bacteria or a log 4 reduction against yeasts. In fields 2 to 4 there needs to be an average carry-over of less than 50 cfu for each organism.


A number of UK laboratories have now validated this test method and more data and understanding of the test is available. The test method was introduced with the aim of closely simulating practical conditions of application such as contact time, temperature and interfering substances, also including pre-drying specified test organisms onto a test surface and wiping the product onto the test surface with a wipe. The laboratory that pioneered the test method identified key factors which affect wiping efficacy including wipe material, wipe quality, impregnation volume, application pressure and application technique. The intention was that the method would be suitable to differentiate between wipes.
Disinfectant manufacturers were hopeful that the test method would highlight that there is no potential cross contamination caused by using the disinfectant wipe and it would also confirm the compatibility of the wipe substrate and disinfectant active, that we highlighted was a particular issue with our previous test work.
Disinfectant manufacturers were hopeful that the test method would highlight that there is no potential cross contamination caused by using the disinfectant wipe and it would also confirm the compatibility of the wipe substrate and disinfectant active, that we highlighted was a particular issue with our previous test work.
Potential drawbacks with the test
Our initial test work generated some results that we were not expecting and led us to investigate the test in more detail. We also had in depth discussions about the test, with the test house carrying out the work (MGS Laboratories in Gosport, UK). Things to be aware of when sending product for testing against EN16615 are:
Unless the manufacturer or facility specifies the wipe to be used with a fluid sent for test, the wipe will be a standard industrial wipe which will not behave in the same way as a cleanroom wipe. “5.3.2.17 Tools for mechanical action, wipe of 17,5 cm x 28 cm, Composition: 55 % pulp, 45 % Polyethylenterephthalat (PET); (eg Tork Premium Spezial Tücher)”.
If not specified the wipe will be wrapped in a single layer around the granite block, this may not be the way the wipe is used in your cleanroom.
Unless the manufacturer or facility specifies the wipe to be used with a fluid sent for test, the wipe will be a standard industrial wipe which will not behave in the same way as a cleanroom wipe. “5.3.2.17 Tools for mechanical action, wipe of 17,5 cm x 28 cm, Composition: 55 % pulp, 45 % Polyethylenterephthalat (PET); (eg Tork Premium Spezial Tücher)”.
If not specified the wipe will be wrapped in a single layer around the granite block, this may not be the way the wipe is used in your cleanroom.

If not specified regardless of size or substrate of the wipe, 16 ml fluid will be used to saturate the wipe. This can have a significant effect on the results. Some of the initial failures seen at the lab were the result of insufficient fluid being released from the wipe either because the wipe was inadequately saturated, or the wipe does not want to give up the fluid. Many wipes saturated with cleaning in mind will only release a small amount of fluid to the surface, as a wipe optimised for particle pick up is saturated just enough to break the binding layer of fluid between a particle and the surface and not so wet that it releases the particle back to the surface again. Wipes optimised for surface disinfection need to be saturated enough so they don’t become unmanageable with drips but can lay down a visible film of disinfectant for the contact time required.
The substrate of the wipe also has an effect on how fluid is absorbed and released to the surface. Organic materials such as cotton and cellulose are able to absorb fluid into the hydrophilic fibre itself whilst man-made materials such as polyester, adsorb liquids into the interstitial spaces between the fibres. In general, synthetic wipes (polyester and polypropylene) tend to be more sorbent as the fibre size is reduced, with microfibre products being the most sorbent option. Manmade fibres such as polypropylene are ideal for laying down a metered release of solvent.
The test does not look at how much fluid the wipes releases to the surface. This could be easily resolved by weighing the wetted wipe before and after the test.
There is no detailed description or validation in the test method of the swabbing technique to be used for recovery of the organisms from the test squares. The standard simply states that the whole surface of each test field should be rubbed with two swabs, one dry and then wet, then swabbed again with a dry swab until the test field is visibly dry. The whole procedure should take no more than 1 min per test field. This is probably the area which will lead to the largest variation between labs, if the swab recovery is carried out differently. An option for improvement could be to validate the swab recovery phase by checking with contact plates.
The influence of mechanical action
As a wipes manufacturer we were less certain when the test was announced that it would be the definitive answer to testing presaturated disinfectant wipes as our in-house testing of wipes had always indicated that significant log reductions in viable organisms could be gained simply by the mechanical action of a wipe or mop. This is especially true of microbfibre fabrics which are ideal at picking up microscopic particles.

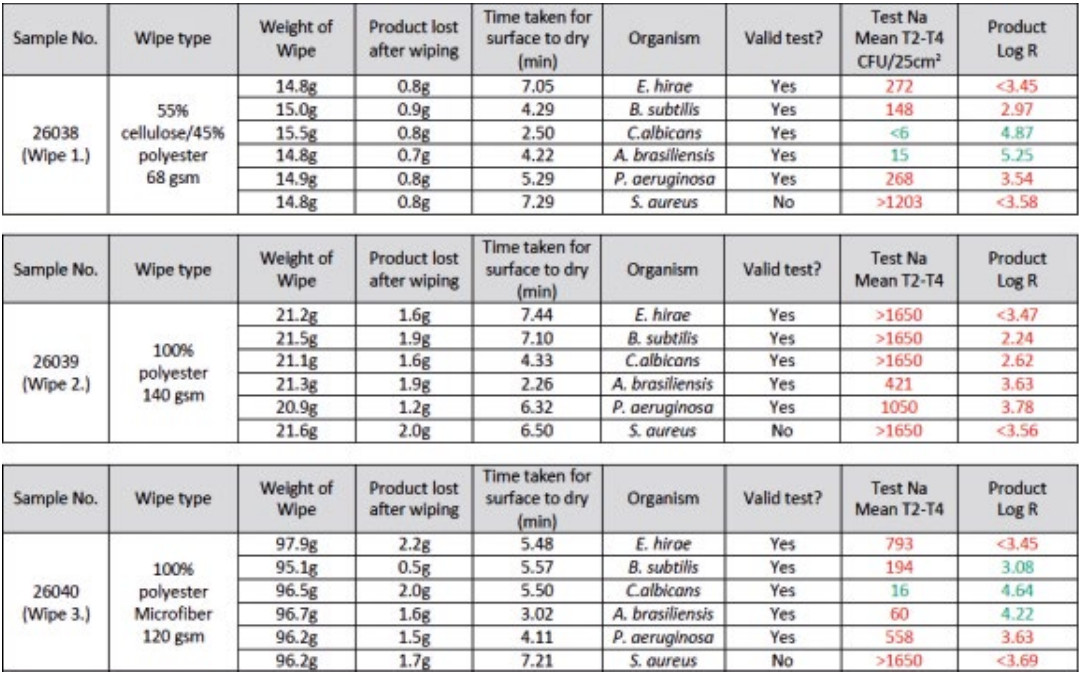
We were so confident in this mechanical action of the wipes that instead of repeating the testing on the disinfectant wipes that we had previously tested we decided to see what results we could achieve with three different wipes substrates simply saturated with purified water. Three different substrates of cleanroom wipe were chosen, 68 gsm polyester/cellulose blend, 140 gsm knitted polyester wipe and a 120 gsm 100 % polyester microfibre wipe, all of which would be commonly used in different grades of cleanroom. As the wipes are different sizes and weights they were saturated with differing amounts of water, to ensure good layout of fluid, namely 12 ml, 15 ml and 60 ml respectively.
They were then tested against EN16615 test with the addition of A. Brasiliensis as a test organism. The summary results from this testing can be seen in Table 2. Two of the substrates tested passed the EN16615 test against C. Albicans and one substrate also passed against A. Brasiliensis.
If the test method is capable of highlighting compatibility issues between the wipe and the disinfectant all of the test results should have been a fail as there was no microbiological activity on the wipe. Interestingly one of the harder to kill organisms in the test{11] passed when using the polyester/cellulose wipe. The yeast passed with both the polyester wipe and the polyester/cellulose wipe. This clearly highlights how significant mechanical action is in the wiping process and the ability of different wipes substrates to pick up and retain particles/microorganisms.
If the results were presented in their entirety as above then it would be clear that there is something odd with the results and a conclusion would be drawn that the “disinfectant” was not hugely efficacious and probably mechanical action was helping to achieve the results. The concern is if the test had been carried out solely on the yeast or fungal organisms to show efficacy specifically against those organisms, maybe because a marginal fail had been achieved on a surface test (EN13697) then this would have misrepresented the efficacy of the disinfectant.
There is an argument to say that the test validates the process which is being carried out, the effectiveness of the fluid to kill and the substrate to pick up and retain particles. However, it is not accurate to suggest the test confirms compatibility between the wipe and the disinfectant as mechanical action could mask any potential incompatibilities. Also, what cannot be deduced from the test is whether a wipe with no microbial efficacy does release the particles/microorgansims at some point. Great care would have to be taken to ensure wipes were only used for a validated amount of time, placed directly into waste receptacles. There could still be a possibility of organism release as a wipe was folded and refolded for example.
They were then tested against EN16615 test with the addition of A. Brasiliensis as a test organism. The summary results from this testing can be seen in Table 2. Two of the substrates tested passed the EN16615 test against C. Albicans and one substrate also passed against A. Brasiliensis.
If the test method is capable of highlighting compatibility issues between the wipe and the disinfectant all of the test results should have been a fail as there was no microbiological activity on the wipe. Interestingly one of the harder to kill organisms in the test{11] passed when using the polyester/cellulose wipe. The yeast passed with both the polyester wipe and the polyester/cellulose wipe. This clearly highlights how significant mechanical action is in the wiping process and the ability of different wipes substrates to pick up and retain particles/microorganisms.
If the results were presented in their entirety as above then it would be clear that there is something odd with the results and a conclusion would be drawn that the “disinfectant” was not hugely efficacious and probably mechanical action was helping to achieve the results. The concern is if the test had been carried out solely on the yeast or fungal organisms to show efficacy specifically against those organisms, maybe because a marginal fail had been achieved on a surface test (EN13697) then this would have misrepresented the efficacy of the disinfectant.
There is an argument to say that the test validates the process which is being carried out, the effectiveness of the fluid to kill and the substrate to pick up and retain particles. However, it is not accurate to suggest the test confirms compatibility between the wipe and the disinfectant as mechanical action could mask any potential incompatibilities. Also, what cannot be deduced from the test is whether a wipe with no microbial efficacy does release the particles/microorgansims at some point. Great care would have to be taken to ensure wipes were only used for a validated amount of time, placed directly into waste receptacles. There could still be a possibility of organism release as a wipe was folded and refolded for example.
Table 2:
A simple way to check whether mechanical action is masking any incompatibility issues would be to check the wipe after testing for the amount of micobial contamination in the wipe. Alternatively, the test should only be used as part of a jigsaw of tests. The validation of an efficacious disinfectant should include a pass on a surface without mechanical action (EN13697), a pass using EN16615 and the cleanroom wipes to be used and in the absence of any published Phase 3 tests, the final piece of the jigsaw ongoing environmental testing. A presaturated wipe could be tested against EN16615 with the extra step of testing the wipe to see if microbial contamination has been killed on the wipe itself or coupled with a test of the disinfectant squeezed from the wipe and then tested against EN13697. This would confirm what is contributing to the overall result, efficaciousness of the disinfectant and the additional reduction from good mechanical pick up from the wipe.
Summary
With the increased prevalence of presaturated wipes in the cleanroom environment and new guidelines requesting the use of presaturated sporicidal wipes[12] a robust EN test for the testing of disinfectant presaturated wipes was definitely needed. However, due to the significant log reductions which can be achieved with wet wiping alone further consideration needs to be given improving the test method and care needs to be taken when reading results in exclusion.
Bibliography
[1] MHRA Questions and Answers for Specials manufacturer’s Jan 2015 update
[2] Guidance for Aseptic Transfer Processes in the NHS: Addressing sporicidal issues July 2015 Edition 1 NHS Pharmaceutical Quality Assurance Committee by the NHS Pharmaceutical Micro Protocols Group. Lead authors Mark Oldcorne, Tim Sizer
[3] Karen Rossington, When one and one doesn’t equal two. Cleanroom Technology, June 2013
[4] Valerie Williamson Surface Sanitation in Healthcare: Why your disinfection system might be letting you down. Infection Control Today Vol 11 No 8 Aug 2007
[5] R Bloss, S Meyer, G Kampf Adsorption of active ingredients of surface disinfectants depends on the type of fabrics used for surface treatments Journal of Hospital Infection Iss 75 Nov 2010
[6] Mac Dougall and Morris, Infection Control Today, June 2006
[7] Ecolab White Paper
[8] Contec Technical Note TN0515 Comparison of presaturated wipes against EN standard efficacy tests.
[9] BS EN 1276:1997 Chemical Disinfectants and Antiseptics — Quantitative suspension test for the evaluation of bactericidal activity of chemical disinfectants and antiseptics used in food, industrial, domestic and institutional areas.
[10] BS EN 1650:1998 Chemical Disinfectants and Antiseptics — Quantitative suspension test for the evaluation of fungicidal activity of chemical disinfectants and antiseptics used in food, industrial, domestic and institutional areas.
[11] BS EN 13704:2002 Chemical Disinfectants and Antiseptics — Quantitative suspension test for the evaluation of sporicidal activity of chemical disinfectants used in food, industrial, domestic and institutional areas.
[12] S.A. Sattar, C. Bradley, R. Kibbee, R. Wesgate, M.A.C. Wilkinson, T. Sharpe , J-Y. Maillard Disinfectant wipes are appropriate to control microbial bioburden from surfaces: use of a new ASTM standard test protocol to demonstrate efficacy. Journal of Hospital Infection 91 (2015) 319 – 325
[13] BS EN 16615:2015 Chemical disinfectants and antiseptics. — Quantitative test method for the evaluation of bactericidal and yeasticidal activity on non-porous surfaces with mechanical action employing wipes in the medical area (4-field test). Test method and requirements (phase 2, step 2)
[1] MHRA Questions and Answers for Specials manufacturer’s Jan 2015 update
[2] Guidance for Aseptic Transfer Processes in the NHS: Addressing sporicidal issues July 2015 Edition 1 NHS Pharmaceutical Quality Assurance Committee by the NHS Pharmaceutical Micro Protocols Group. Lead authors Mark Oldcorne, Tim Sizer
[3] Karen Rossington, When one and one doesn’t equal two. Cleanroom Technology, June 2013
[4] Valerie Williamson Surface Sanitation in Healthcare: Why your disinfection system might be letting you down. Infection Control Today Vol 11 No 8 Aug 2007
[5] R Bloss, S Meyer, G Kampf Adsorption of active ingredients of surface disinfectants depends on the type of fabrics used for surface treatments Journal of Hospital Infection Iss 75 Nov 2010
[6] Mac Dougall and Morris, Infection Control Today, June 2006
[7] Ecolab White Paper
[8] Contec Technical Note TN0515 Comparison of presaturated wipes against EN standard efficacy tests.
[9] BS EN 1276:1997 Chemical Disinfectants and Antiseptics — Quantitative suspension test for the evaluation of bactericidal activity of chemical disinfectants and antiseptics used in food, industrial, domestic and institutional areas.
[10] BS EN 1650:1998 Chemical Disinfectants and Antiseptics — Quantitative suspension test for the evaluation of fungicidal activity of chemical disinfectants and antiseptics used in food, industrial, domestic and institutional areas.
[11] BS EN 13704:2002 Chemical Disinfectants and Antiseptics — Quantitative suspension test for the evaluation of sporicidal activity of chemical disinfectants used in food, industrial, domestic and institutional areas.
[12] S.A. Sattar, C. Bradley, R. Kibbee, R. Wesgate, M.A.C. Wilkinson, T. Sharpe , J-Y. Maillard Disinfectant wipes are appropriate to control microbial bioburden from surfaces: use of a new ASTM standard test protocol to demonstrate efficacy. Journal of Hospital Infection 91 (2015) 319 – 325
[13] BS EN 16615:2015 Chemical disinfectants and antiseptics. — Quantitative test method for the evaluation of bactericidal and yeasticidal activity on non-porous surfaces with mechanical action employing wipes in the medical area (4-field test). Test method and requirements (phase 2, step 2)
ACKNOWLEDGEMENTS
The author would like to acknowledge the input and work carried out by Neil Simpson, R and D, Contec and Kim Morwood, Director MGS Laboratories.
AUTHOR
Karen Rossington
Global Cleanroom Market Manager, Contec Inc.
Global Cleanroom Market Manager, Contec Inc.
CONTACT
Contec, Vannes, France
Tel.: +33 2 97 43 76 98
infoeu@contecinc.com
www.contecinc.com
Tel.: +33 2 97 43 76 98
infoeu@contecinc.com
www.contecinc.com



